
An Experiment on a Bird in the Air Pump by Joseph Wright of Derby, 1768

The Mouse’s Petition by Francesco Bartolozzi, 1791
When asked if I would incorporate the Periodic Table into my classes as part of an institute-wide celebration of the International Year of the Periodic Table, I eagerly undertook the challenge of designing an ENGL 1102 course considering 18th-century rhetorics of science for Summer 2019. I also decided to include scientific correspondence in my Spring 2019 course on 18th-century letter culture. While brainstorming possible assignments and readings, I turned to correspondence by Joseph Priestley, an eighteenth-century chemist whose work on combustion and the creation of carbonated water I thought would interest my students curious about science and technology – as many of our Georgia Tech students are. Priestley also has claim to fame in his use of mice as test subjects, the inspiration for Anna Laetitia Barbauld’s poem “The Mouse’s Petition.” I ended up selecting letters from Priestley to Josiah Wedgewood, Benjamin Franklin, and other men in the Royal Society and its Birmingham off-shoot, the Lunar Society, including engineer James Watt. Eighteenth-century nomenclature for geologic specimens and scientific apparati peppered the pages of Priestley’s letters. My students would read and discuss these letters in my Spring class, which would give them the opportunity to reflect on the progress scientific methods have made (or not) in the last two centuries.
After the engagement my Spring students showed with Priestley’s letters, I started to see the shape of my Summer 2019 course, which I titled “Invention, Exploration, and the Elements: The Rhetoric of Science from the 18th Century to Today.” The purpose of the course would be to acknowledge eighteenth-century science as foundational for ongoing contemporary innovation, but I also wanted to emphasize how, paired with digital strategies, learning to communicate about science from the past could help my students successfully convey research findings in their own future careers.

The cover of The Disappearing Spoon by Sam Kean, 2010
Crucial to my course’s development was Sam Kean’s book on the construction of the Periodic Table, The Disappearing Spoon. I wanted to expand my use of the Table in the course structure and, given my own research focus, I decided to integrate Kean’s narrative with genres of eighteenth-century science communication. Kean’s text draws on anecdotes and historical accounts to convey how each element on the Periodic Table was discovered and by whom, including cameos by several eighteenth-century figures such as Priestley, Antoine LaVoisier, and Johann Friedrich Böttger. Kean tracks the development of the table as we know it today as a process of trial and error, of thoughtful and purposeful design. The composition of the table was surprisingly speculative, but ultimately rooted in an understanding of organizational principles that require precise calculation, prediction, and measurement to achieve. Such principles of process-based composition are in line with the foci of the Writing and Communication Program’s WOVEN curriculum, particularly given the multimodal nature of the periodic table, which conveys information via visual and textual cues resulting from deliberate design choices. Integrating The Disappearing Spoon with eighteenth-century texts about discovery, the natural world, and scientists seemed to be a natural progression.
Course Overview
Throughout the Summer 2019 term my students read The Disappearing Spoon as well as a range of eighteenth-century primary sources and texts. Such texts spanned multiple genres, including correspondence like Priestley’s letters; poetry, including Barbauld’s “The Mouse’s Petition” and James Thomson’s “To The Memory of Sir Isaac Newton;” fiction excerpts from Mary Shelley’s Frankenstein and a graphic novel adaptation of Shelley’s original; broadsides such as Edmund Halley’s predictions for the 1715 solar eclipse; and articles from eighteenth-century volumes of Philosophical Transactions, the journal of the Royal Society that is still publishing articles today. For these readings, we considered rhetorical situation, context, rhetorical appeals, and how each text made meaning as it engaged with “natural philosophy,” the eighteenth-century ubiquitous term for science.
Artifact Design Sequence
Engaging with videos, digitally-designed posters, and digital media webtext in the form of multimodal blog posts, my students practiced principles of design with alphanumeric text, images, audio, and film. The assignment sequence I constructed challenged them to discover details about an eighteenth-century scientist’s life and work and an eighteenth-century scientific discovery, before culminating in a collaborative periodic table of the students’ own design. Each artifact required several process document deliverables as the students worked through creating the final project. Students peer reviewed each other’s work, collaborating on design through feedback and reflection, much as they might do in their computer science capstone course, their business communication course, or their aerospace or chemistry labs in the future.
Work by Angel Hsieh
In keeping with Kean’s text, which considers the individual narratives of people who discovered the chemical elements as well as the design and organization of the Periodic Table itself, I designed our assignment sequence to mirror the historical synergy between scientists, discoveries, and organizational design principles. Beginning with multimodal biographical blog posts and then shifting to short videos about scientific discoveries, students practiced research strategies and digital design by choosing their topics as well as their audiences. For their final artifact, students collaboratively designed their own periodic tables of “elements,” where they decided what “elements” to organize, determined criteria for grouping, chose the shape and structure of their tables, selected colors and images to represent their “elements,” and digitally constructed their tables through a drafting and revision process.
Work by Caroline Adkins
Artifact 1: Biographical Blog Post
Reading through Kean’s text, students were surprised to learn how often women were involved in scientific advancements, and how often discoveries affected people’s personal lives. For example, Kean notes that Marie Curie’s, Clara Immerwahr’s, and Maria Goeppert-Mayer’s private lives and relationships with their husbands affected their participation in research as much if not more than general prejudices against women in the field. Artifact 1 asked students to explore the people behind the scientific discoveries of the eighteenth century by researching the life and work of one scientist and creating a biography of that person on a collaborative WordPress blog I set up for the class.
After choosing their scientists, students designed their posts while also remaining conscious of the affordances of the blog as a web medium. They used each of the WOVEN modes to construct their blog posts, purposefully embedding appropriate video clips into their prose, and wrapping text around images, memes, and gifs. Two students wrote for younger audiences, specifically addressing their biographies to kids (see Angel Hsieh’s “Joseph Priestley For Kids” above). Some students highlighted the fact that women scientists like Caroline Herschel contributed significantly to their male counterparts’ work or were the ones actually making the discoveries, despite the credit going to men. One student found that a woman designed a corn mill that produced that perennial Southern favorite, grits, but her husband held the patent because women were legally prohibited from doing so. Another student described how a Japanese doctor, Seishū Hanaoka, used anaesthesia in the 1760s, specifically for breast cancer surgery. These students’ blog posts deconstruct the dominant narrative of Western science conducted by a white man in a laboratory, emphasizing instead discoveries produced in Asia and by a woman that had wide-ranging benefits and applications, even today.
Work by Chloe Devre
Artifact 2: Short Documentary Video
After blog posts that discussed the people behind the research, Artifact 2 focused on the discoveries themselves, challenging students to create a 4-6 minute video conveying information about an eighteenth-century scientific innovation. Kean’s argument in The Disappearing Spoon is that the creation of the Periodic Table and the discovery of 118 elements was not achieved in isolation and relied on knowledge gained through painstaking processes developed over many years. Following Kean’s model, many of my students asserted that information in a variety of subjects had its roots in eighteenth-century natural philosophy.
For their videos, students constructed an argument about their particular discovery, compiled video clips and images as well as music and sound effects to use in their film, and recorded audio files to use as narration. Several students imitated similar styles of informational video by incorporating humor and classroom instruction processes. The results were fantastic. They traced the history of work on electromagnetism to André-Marie Ampère, and nuclear reactor technology to James Watt’s improvements to the steam engine. As part of an argument detailing alchemy as proto-chemistry in an imitation of “World History: Crash Course,” one student discussed Johann Böttinger’s false claim of perfecting the transfiguration of silver into gold, which ultimately led to his discovery of a technique to create porcelain. In each of these examples, students showcased how familiar things or ideas, such as fine china or the refraction of sunlight in the atmosphere, were not based on recent science but instead grew out of experimentation – or just plain fabrication – conducted more than two centuries ago. The video medium allowed students to build on skills utilized in designing Artifact 1. They were obliged to expand the amount and types of knowledge they conveyed, combining image and written text with deliberate choices in building a soundscape to emphasize specific concepts in their arguments.
Work by Diana Kim
Artifact 3: Collaborative Periodic Table

Work by Carson Clements, Natalie Raia, and two other classmates
The culminating artifact for my ENGL 1102 summer session course was the Periodic Table of “elements.” I challenged my class to collaboratively create their own versions of the Periodic Table by incorporating principles of visual design, considering how to signify meaning using symbols, and curating each example or unit that made up the whole with deliberate and thoughtful choices.
Just as Kean argues that the Periodic Table’s structure relies on logical rationales, each table my students created highlighted specific justifications for their elements’ symbolism and placements within the cohesive structure. One group relied on the fact that atomic mass doesn’t factor for the wands of Harry Potter, but wood and core materials do. Another group acknowledged that electron bonding properties are important for arranging calcium, nitrogen, or uranium on the Periodic Table, but not for classifying various potato preparation methods; however, considering potato chips as a division much like lanthanides on the actual Table achieved the effect of sectioning off a larger portion of fried potato in their Periodic Table of the Pomme de Terre.
The way the tables look as holistic visual documents matters as much as any single image or alphanumeric text utilized within the tables. The Fruity Table, for example, took the shape of an F and included an image of each fruit element. This particular group anticipated their choice of visual components would provoke instantaneous recognition in their audience and so emphasized that aspect of their design. Background color imparts clear meaning, as does the border for each block, and the attribute of acidity functions as an organizational principle. Though shape and material are not the same, the Fruity Table clearly adapts many of the same principles of the original Periodic Table.
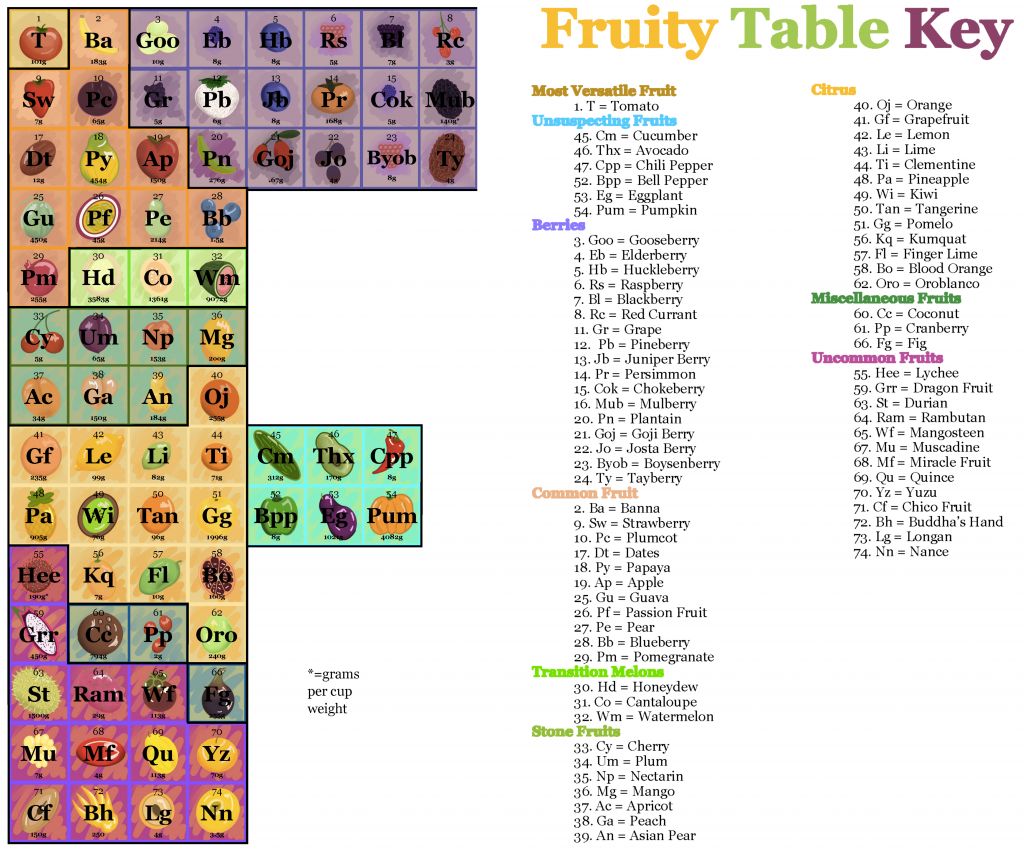
Work by Corinna Alting, Angel Hsieh, Diana Kim, Asia Taylor, and another classmate
Reflection
Just like with any experiment, some surprises arose from my assignments. My general emphasis on allowing students to choose their own rhetorical situation with regard to audience, style, and tone can occasionally lead to some variations in the work my students produce, limitations of time, equipment, and skill in technology aside. In the case of the Periodic Table of the Pomme de Terre, my students surprised me with their creativity. This particular group created two versions: one static and one in Prezi form, which allowed them to embed videos and incorporate multimodality in ways I had not expected when I designed the assignment. I had anticipated digitally-designed posters, reminiscent of the actual Periodic Table; the Prezi Periodic Table of the Pomme de Terre provided a level of interaction beyond my original conception of what my students would produce. The Prezi form changes the dynamics of how the table can be read so that when I can click through the table to access details, I can also read it forwards, backwards, or by randomly selecting different parts. I can rewatch the embedded video material, and I can delve more deeply into information in a way the standard table limits. With the static form I can learn more about the concepts presented, but I would need to have access to other texts or electronic resources. Though I have incorporated interactivity into other assignments (a board game project in Spring 2018 and a group mapping artifact in Spring 2019, for example), I had not specifically asked for it in the Periodic Table assignment. The Pomme de Terre group’s success in integrating interactive elements into their artifact inspires me to consider including an interactive component requirement for future assignments.
Likewise, the success I had in designing this course around the Periodic Table of Elements was pleasantly surprising, though not completely unexpected, given Georgia Tech students’ general enthusiasm for all things science and technology. They were surprised to hear the topic of the course, but overall, the connections we were able to make between 18th-century scientific thought and contemporary STEM subjects which they might engage in the near future convinced them of the importance of understanding how far scientific advancements have come and where they might be going. The grounding the students gained in considerations of rhetorics of science highlighted for them how the environment for communication has changed and yet remains similar. Tables, charts, and diagrams remain important multimodal communication genres. Discourse between scientists and engineers may occur much more quickly in the 21st century than in the 18th, thanks to electricity, cars, and the internet, but the exchange that occurs when work is published, ideas are discussed, and prior work is built upon remains as vital to the advancement of knowledge today as it did 250 years ago.
When we discussed Halley’s predictions for early 18th-century solar eclipses, I was reminded of the solar eclipse in 2017. We may not attach political or cultural importance to astronomical events today in the way Britons did in 1715, but the ephemera that record such events with words and images highlight how the construction and communication of knowledge has benefited humanity throughout our history.
To read more about my course, please visit the College of Science’s article featuring interviews with several students.
